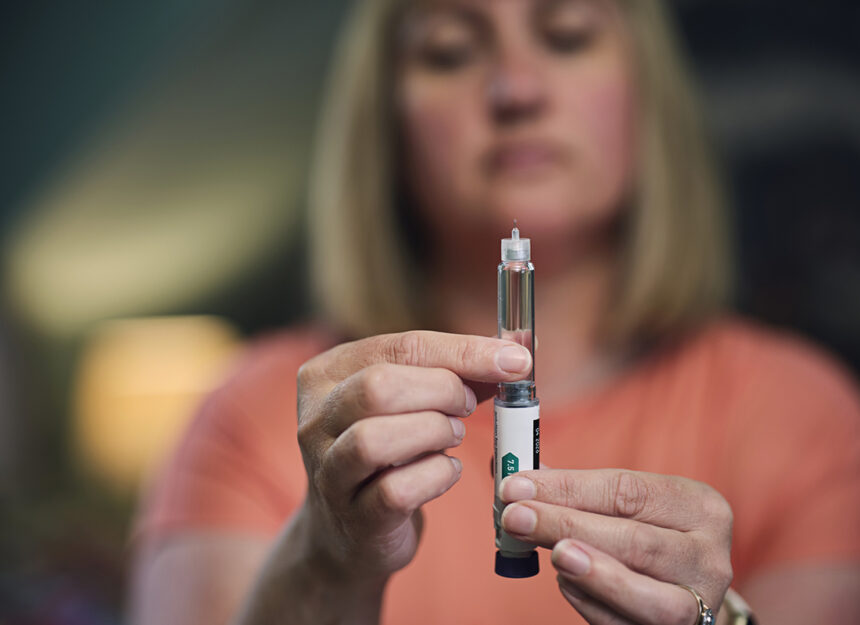Key Takeaways
• EMA approves higher semaglutide dose for obesity ||| • STEP UP trial: 20.7% weight loss with 7.2 mg ||| • Safety profile of higher dose similar ||| • Wegovy's global regulation in progress.
Recommendation from EMA Moves Higher Dose closer to Approval
The European Medicines Agency recently gave their approval of using a higher dose of semaglutide for obesity treatment, opening up more treatment options throughout Europe. Their recommendation concerned Wegovy(r), with final authorization ultimately resting with the European Commission following this guidance from EMA; should approval come through, manufacturer anticipates new dose to become available within days across Europe as an application was also submitted specifically designed to handle such higher strength doses.
STEP UP Trial Results Demonstrate Greater Weight Loss Success
The European Medicines Agency’s assessment is grounded on robust clinical data from the STEP UP program. A 72-week late stage trial with over 1,400 adults suffering from obesity but without diabetes found those receiving semaglutide at 7.2 mg achieved an average weight loss of 20.7%; by comparison, those on semaglutide at its currently approved dosage (2.4 mg) only lost 17.5% on average; one third lost 25+% from their original bodyweight while body composition analyses demonstrated 84% came from fat mass reduction while muscle function remained intact during that trial period.
Safety Profile and Additional Health Benefits
Trial data has demonstrated the safety profile of higher doses of semaglutide is comparable to lower dosages; nausea and diarrhea were among the most frequently reported side effects, followed by weight reduction benefits like decreased cardiovascular events such as heart attacks and strokes as well as helping ease knee osteoarthritis pain associated with obesity. Studies also confirmed benefits in terms of knee osteoarthritis pain relief – another side benefit associated with weight reduction.
Global Review and Clinical Considerations
Regulation for Wegovy(r), with 7.2 mg dosage strength is currently underway in both the US, UK, and other countries, following submission to U.S. regulators under an expedited review process. Not intended as an initial treatment but studied specifically among patients already taking semaglutide who require further appetite suppression, this higher-dose Wegovy could represent a meaningful advance in obesity management, providing clinicians and patients alike greater flexibility as they pursue deeper weight loss through clinical significance weight management.